Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 CsI (v = 0, j = 0) +
CsI (v = 0, j = 0) +  Cs
Cs 
 Cs + I
Cs + I Cs
CsKobayashi, Takanori*; Matsuoka, Leo*; Yokoyama, Keiichi
Nihon Enerugi Gakkai-Shi, 96(10), p.441 - 444, 2017/10
To investigate the reaction cross section of the cesium exchange reaction of  CsI (v = 0, j = 0) +
CsI (v = 0, j = 0) +  Cs
Cs 
 Cs + I
Cs + I Cs, we performed quasi-classical trajectory calculations on the potential energy surface calculated by the ab initio molecular orbital theory. The potential energy surface shows that intermediate Cs
Cs, we performed quasi-classical trajectory calculations on the potential energy surface calculated by the ab initio molecular orbital theory. The potential energy surface shows that intermediate Cs I is formed without entrance barrier and has two equivalent Cs-I bonds. The reaction cross sections decrease monotonically with increasing collision energy. The rate constant k (v = 0, j = 0) was estimated to be about 3
I is formed without entrance barrier and has two equivalent Cs-I bonds. The reaction cross sections decrease monotonically with increasing collision energy. The rate constant k (v = 0, j = 0) was estimated to be about 3 10
10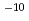 cm
cm molecule
molecule s
s at temperatures ranging from 500 to 1200K and a slight negative temperature dependence was observed.
at temperatures ranging from 500 to 1200K and a slight negative temperature dependence was observed.
Kubo, Hitoshi*; Oshima, Yusuke*; Iwai, Yasunori
JETI, 63(10), p.33 - 36, 2015/09
Tanaka Kikinzoku Kogyo provides a broad range of precious metals products and technologies. Tanaka Kikinzoku Kogyo and Japan Atomic Energy Agency have jointly developed a new method of manufacturing catalysts involving hydrophobic processing with an inorganic substance base. As a result, previous technological issues were able to be solved with the development of a catalyst that exhibited no performance degradation in response to radiation application of 530 kGy, a standard for radiation resistance, and maintenance of thermal stability at over 600 C, which is much higher than the 70
C, which is much higher than the 70 C temperature that is normally used. The application of this catalyst to the liquid phase catalytic exchange process is expected to overcome significant technological hurdles with regards to improving the reliability and efficiency of systems for collecting tritium from tritiated water. It is also anticipated that the hydrophobic platinum catalyst manufacturing technology used for this catalyst could be applied to a wide range of fields other than nuclear fusion research. It was verified that if applied to a hydro oxidation catalyst, hydrogen could be efficiently oxidized, even at room temperature. This catalyst can also contribute to improving safety at non-nuclear plants that use hydrogen in general by solving the aforementioned vulnerability issue.
C temperature that is normally used. The application of this catalyst to the liquid phase catalytic exchange process is expected to overcome significant technological hurdles with regards to improving the reliability and efficiency of systems for collecting tritium from tritiated water. It is also anticipated that the hydrophobic platinum catalyst manufacturing technology used for this catalyst could be applied to a wide range of fields other than nuclear fusion research. It was verified that if applied to a hydro oxidation catalyst, hydrogen could be efficiently oxidized, even at room temperature. This catalyst can also contribute to improving safety at non-nuclear plants that use hydrogen in general by solving the aforementioned vulnerability issue.
Isobe, Kanetsugu; Nakamura, Hirofumi; Kaminaga, Atsushi; Higashijima, Satoru; Nishi, Masataka; Konishi, Satoshi*; Nishikawa, Masabumi*; Tanabe, Tetsuo*
Fusion Science and Technology, 48(1), p.302 - 305, 2005/07
Times Cited Count:5 Percentile:35.8(Nuclear Science & Technology)no abstracts in English
Katayama, Kazunari*; Takeishi, Toshiharu*; Nagase, Hiroyasu*; Manabe, Yusuke*; Nishikawa, Masabumi*; Miya, Naoyuki; Masaki, Kei
Fusion Science and Technology, 48(1), p.561 - 564, 2005/07
Times Cited Count:1 Percentile:10.45(Nuclear Science & Technology)no abstracts in English
Nishikawa, Masabumi*; *; Kawamura, Yoshinori; Nishi, Masataka
JAERI-Conf 98-006, p.170 - 182, 1998/03
no abstracts in English
 ZrO
ZrO packed bed
packed bedKawamura, Yoshinori; Enoeda, Mikio; Okuno, Kenji
Fusion Engineering and Design, 39-40, p.713 - 721, 1998/00
Times Cited Count:8 Percentile:57.28(Nuclear Science & Technology)no abstracts in English
Nishikawa, Masabumi*; Baba, Atsushi*; *; Kawamura, Yoshinori
Fusion Engineering and Design, 39-40, p.615 - 625, 1998/00
Times Cited Count:14 Percentile:73.06(Nuclear Science & Technology)no abstracts in English
Baba, Atsushi*; Nishikawa, Masabumi*; Kawamura, Yoshinori; Okuno, Kenji
Journal of Nuclear Materials, 248, p.106 - 110, 1997/09
Times Cited Count:4 Percentile:37.13(Materials Science, Multidisciplinary)no abstracts in English
Kawamura, Yoshinori; Nishikawa, Masabumi*; *; Okuno, Kenji
Journal of Nuclear Materials, 230, p.287 - 294, 1996/00
Times Cited Count:43 Percentile:94.61(Materials Science, Multidisciplinary)no abstracts in English
 O(VOM-21H); Tritium Chemical Form as a Function of Sweep Gas Composition
O(VOM-21H); Tritium Chemical Form as a Function of Sweep Gas Composition; ; Kurasawa, T.; ;
JAERI-M 86-130, 20 Pages, 1986/09
no abstracts in English
Kurasawa, T.; ; ; ; ;
JAERI-M 84-087, 55 Pages, 1984/05
no abstracts in English
*; *;
J.Chem.Eng.Jpn., 16(5), p.370 - 377, 1983/00
Times Cited Count:24 Percentile:83.93(Engineering, Chemical)no abstracts in English
; Naruse, Yuji
Nucl.Technol./Fusion, 3, p.112 - 120, 1982/00
no abstracts in English
Nakashima, Mikio; Tachikawa, Enzo; Saeki, Masakatsu;
Journal of Inorganic and Nuclear Chemistry, 43(2), p.369 - 373, 1981/00
Times Cited Count:8 Percentile:37.91(Chemistry, Inorganic & Nuclear)no abstracts in English
; ; Motoishi, Shoji; ;
JAERI-M 5320, 31 Pages, 1973/07
no abstracts in English
Bunseki Kagaku, 14(2), P. 138, 1965/00
no abstracts in English
Watanabe, Masashi; Kato, Masato; Sunaoshi, Takeo*
no journal, ,
The oxygen self-diffusion coefficients in mixed oxide (MOX) fuel are successfully measured using an isotope exchange method under H /H
/H O atmosphere. These results are discussed with respect to the activation enthalpy of lattice defects.
O atmosphere. These results are discussed with respect to the activation enthalpy of lattice defects.

Watanabe, Masashi; Sunaoshi, Takeo*; Kato, Masato
no journal, ,
no abstracts in English